Projects
Epilepsy and dendritic excitability
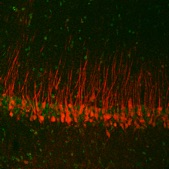
We are studying the phosphorylation-dependent early changes in HCN channel trafficking and how these contribute to the development of epilepsy after neural injury. We are also characterizing human mutations in the HCN1 channel, and how these produce changes in Ih (the current mediated by HCN channels) leading to catastrophic infantile epilepsy and developmental regression.
Funded by the National Institutes of Health.
Cellular mechanisms of antiepileptic drug synergy
This project will attempt to replicate in an animal model the human clinical finding that the combination of lamotrigine and valproate exhibits pharmacodynamic synergy in refractory epilepsy patients. It will then explore underlying cellular mechanisms of this synergism.
Funded by the National Institutes of Health.
Novel biomarkers in refractory epilepsy
We are seeking to identify patterns of abnormal phosphorylation of ion channel proteins in tissue from human patients and in an animal model of refractory epilepsy so to characterize possible novel biomarkers of the epileptogenic zone where seizures arise.
Funded by the UW Royalty Research Fund.
Large-scale analysis of antiepileptic drug efficacy in Lennox-Gastaut Syndrome
We are employing similar techniques to those we used in previous studies that discovered the unique efficacy of the lamotrigine/valproate drug combination in refractory efficacy to determine whether there are effective antiepileptic drug combinations in LGS.
Funded by the Lennox-Gastaut Syndrome Foundation and with the help of the International Seizure Diary Consortium and SeizureTracker.